Abstract
Background: Activating mutations in MYD88 and CXCR4 are present in 95-97% and 30-40% of patients with Waldenström Macroglobulinemia (WM), respectively. Gene expression and transcriptome studies have shown that BCL2 is highly expressed in WM cells, particularly in those patients with activating MYD88 mutations (Chng et al, Blood 2006; Hunter et al, Blood 2016). Use of the BCL2 inhibitor venetoclax triggered apoptosis of WM cells, including primary WM cells from ibrutinib-naïve and ibrutinib-treated patients. In a Phase I dose finding study, venetoclax showed efficacy in 4 WM patients (Davids et al, JCO 2017). We therefore initiated a phase II study to evaluate the safety and efficacy of venetoclax monotherapy in previously treated patients with WM (NCT02677324), and determined the impact of prior BTK-inhibitor (BTK-i) therapy and CXCR4 mutation status on treatment response.
Methods: Venetoclax was administered as outpatient therapy, and followed a ramp-up of 200 mg daily on days 1-7, 400 mg daily on days 8-14, then 800 mg daily for maximum of 2 years. Dose reduction for adverse events (AEs) was permitted. Patients were closely monitored for tumor lysis syndrome (TLS) during the first 24 hours of each dose escalation. Response was assessed using modified IWWM-6 criteria. MYD88 and CXCR4 genotyping was performed as before (Xu et al, BJH 2016).
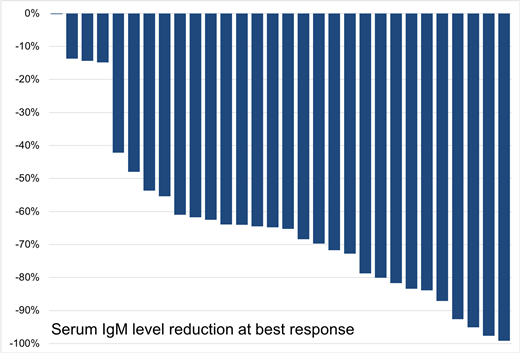
Results: 30 patients with symptomatic WM were enrolled. Their median age was 66 years (range 39-80 years) and 17 (57%) were men. Indications for treatment included constitutional symptoms (52%), anemia (44%), peripheral neuropathy (15%), extramedullary disease (11%) and thrombocytopenia (7%). The median number of prior therapies was 2 (range 1-10), and 15 (50%) patients were previously treated with a BTK-i. All patients carried the MYD88L265P mutation, and 16 (53%) also had a CXCR4 mutation. At baseline, median serum IgM was 3,543 mg/dl (range 642-7,970 mg/dl), median bone marrow involvement was 35% (range 4-95%) and median hemoglobin was 10.6 g/dl (range 6.4-13.5 g/dl). All evaluable patients have completed 6 months of therapy, at a median follow-up of 12 months (range 1-23 months). All patients were successfully escalated to target dose of 800 mg. At 6 months, median serum IgM declined to 1,750 mg/dl (range 49-5,220 mg/dl), median bone marrow involvement declined to 5% (range 0-30%) and median hemoglobin increased to 12.4 g/dl (range 10-15.1 g/dl) (p<0.001 for all variables against baseline). At best response, very good partial response (VGPR) was attained in 5 patients (17%), partial response in 19 (63%), minor response in 2 (7%) and stable disease in 4 (13%), for an overall response rate of 87% and major response rate of 80%. Patients with prior refractory disease showed a lower major response rate versus those with relapsed disease (57% vs. 95%; p=0.01). Moreover, VGPR rates were lower in those with prior BTK-i exposure (7% vs. 27%; p=0.10), and CXCR4 mutations (6% vs. 29%; p=0.10). Median time to response (TTR) was 9 weeks and was slower in patients with prior BTK-i exposure (19 vs. 6 weeks; p=0.02). TTR was not impacted by relapsed vs. refractory disease status nor by CXCR4 mutation status. Two patients progressed at 8 and 10 months on therapy. Overall, treatment was well tolerated and no clinical TLS occurred. Laboratory TLS occurred in one patient with significant extramedullary disease. Grade 4 neutropenia occurred in 4 patients, who responded to G-CSF. Grade 3 AEs included neutropenia (n=7), anemia (n=2), back pain (n=1), constipation (n=1), diarrhea (n=1), headache (n=1), upper respiratory infection (n=1). Grade 2 AEs included anemia (n=5), nausea (n=4), neutropenia (n=3). Grade 1 AEs included nausea (n=9), diarrhea (n=6), rash (n=5). Dose reduction for AEs occurred in two patients (for neutropenia and diarrhea). No instances of IgM flare were observed, and there have been no deaths.
Conclusion: The findings show that venetoclax is well tolerated, and produces high levels of response in patients with symptomatic, previously treated WM, including patients previously exposed to BTK-i. Prior BTK-i exposure and presence of CXCR4 mutations impacted VGPR response attainment.
Castillo:Beigene: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Millennium: Research Funding; Genentech: Consultancy; Abbvie: Consultancy, Research Funding. Allan:Verastem: Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees; Acerta: Consultancy; Genentech: Membership on an entity's Board of Directors or advisory committees; Sunesis: Membership on an entity's Board of Directors or advisory committees. Furman:Loxo Oncology: Consultancy; TG Therapeutics: Consultancy; Verastem: Consultancy; Gilead: Consultancy; Incyte: Consultancy, Other: DSMB; AbbVie: Consultancy; Janssen: Consultancy; Sunesis: Consultancy; Genentech: Consultancy; Pharmacyclics LLC, an AbbVie Company: Consultancy; Acerta: Consultancy, Research Funding. Siddiqi:Juno Therapeutics: Other: Steering committee. Advani:Kyowa: Other: Consulting/Advisory Role; Celgene: Other: Institutional Research Support; Merck: Other: Institutional Research Support; Bristol Myers Squibb: Other: Consultancy/Advisory role and Institutional Research Support; Janssen Pharmaceutical: Other: Institutional Research Support; Regeneron Pharmaceuticals, Inc.: Other: Institutional Research Support; Infinity: Other: Institutional Research Support; Forty Seven, Inc: Other: Institutional Research Support; Gilead/Kite: Other: Consultancy/Advisory Role; Millenium: Other: Institutional Research Support; Agensys: Other: Institutional Research Support; Kura: Other: Institutional Research Support; Roche/Genentech: Other: Consultancy/Advisory Role, Institutional Research Support; Takeda: Other: Consultancy/Advisory Role; Pharmacyclics: Other: Institutional Research Support; Bayer Healthcare Pharmaceuticals: Other: Consultancy/Advisory Role; Cell Medica: Other: Consultancy/Advisory Role; AstraZeneca: Other: Consultancy/Advisory Role; Autolus: Other: Consultancy/Advisory Role; Seattle Genetics: Other: Consultancy/Advisory role, Institutional Research Support. Hunter:Pharmacyclics: Consultancy. Davids:Roche: Consultancy; Surface Oncology: Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astra-Zeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy; MEI Pharma: Consultancy, Research Funding; Astra-Zeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Research Funding; Celgene: Consultancy; Astra-Zeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; MEI Pharma: Consultancy, Research Funding; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Merck: Consultancy; Gilead: Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Surface Oncology: Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Surface Oncology: Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Merck: Consultancy; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Verastem: Consultancy, Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; MEI Pharma: Consultancy, Research Funding; Gilead: Membership on an entity's Board of Directors or advisory committees; BMS: Research Funding; Merck: Consultancy; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sunesis: Membership on an entity's Board of Directors or advisory committees; BMS: Research Funding; Verastem: Consultancy, Research Funding; Celgene: Consultancy; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sunesis: Membership on an entity's Board of Directors or advisory committees; Sunesis: Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Verastem: Consultancy, Research Funding. Treon:Janssen: Consultancy, Other: Travel, Accommodations, Expenses; Pharmacyclics: Consultancy, Other: Travel, Accommodations, Expenses, Research Funding; Johnson & Johnson: Consultancy; BMS: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal